I previously looked at muscle memory on the blog at the beginning of 2019. This time, I’ll be updating the article with new evidence and a more didactic approach.
Want to know what muscle memory is? Don’t miss out!
Index
What it is
Muscle memory is a concept coined by athletes who perceived that after a break (through injury or other reasons) in which muscle mass was lost, they were able to regain their previous muscle volume much faster than when they first started.

Figure I. Kevin Levrone changes. First image: 90s. Second image: 2008. Third image: 2017.
Muscle memory, nothing new on the horizon
The body is efficient, and one of the biological principles that govern life is that efficiency.
The body adapts to an increase in the demands to which we submit it to become more efficient, for example:
When we move to a place that is at a much higher altitude above sea level than we normally are, the partial pressure of oxygen decreases and our body increases the eritropoiesis in order to compensate for the decrease in oxygen uptake from the environment and prevent tissue hypoxia.
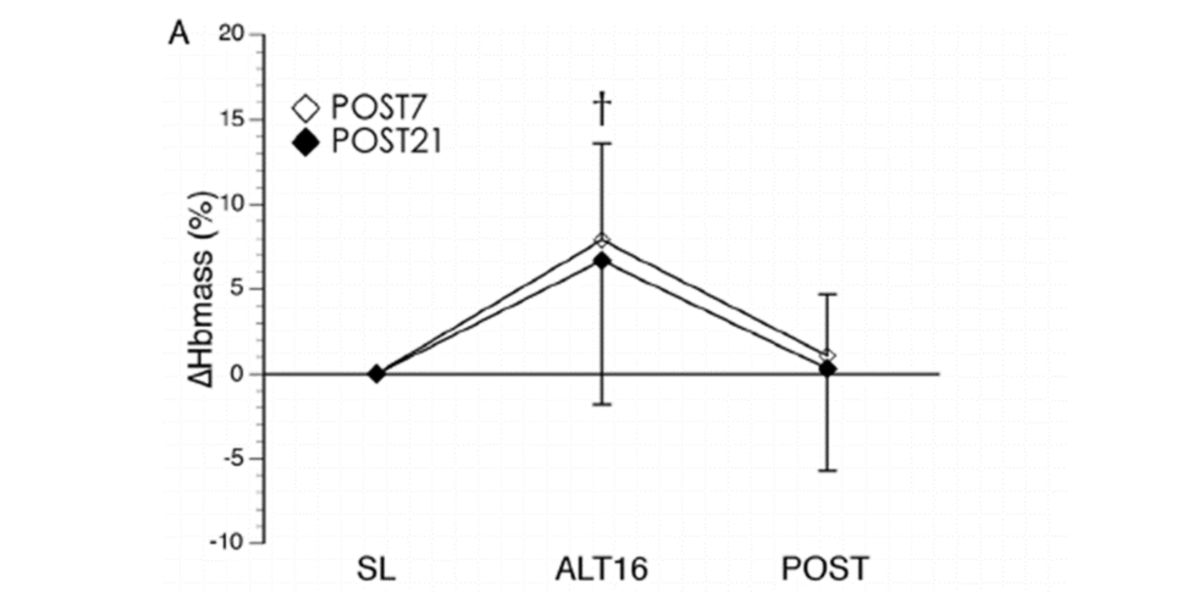
Figure II. Haemoglobin concentration changes after 16 days of acclimatisation at +5000m and after returning to the original level for 7 and 21 days.
When we return to our usual place, the haemoglobin concentrations return to their initial state, because we no longer need that adaptation.
The sae happens with muscle tissue:
In the face of increasing demands on force production, and taking into account that muscle size is one of the major determinants of force production, our tissues adapt to the damage produced by receiving new nuclei that can help repair muscle fibres.
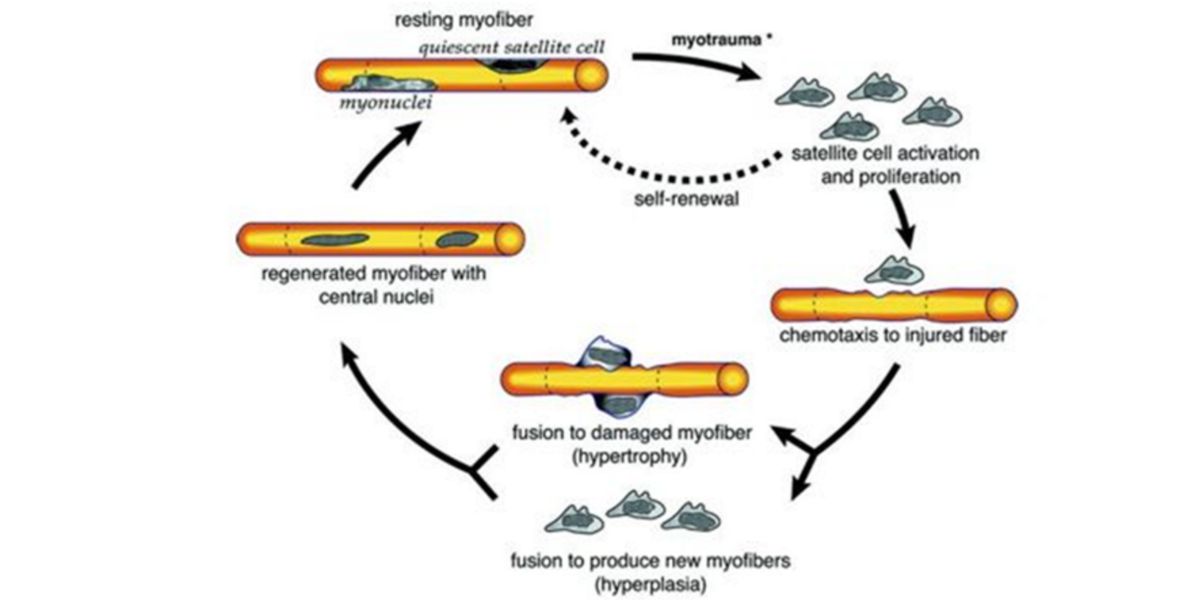
Figure III. Process of proliferation, chemotaxy and fusion of the satellite cells to the damaged muscle fibre.
To understand this process, it’s important to understand that muscle cells are polynuclear cells and that each of these nuclei control the transcriptional activity of a certain area of the muscle (called nuclear domain), let’s imagine it like this:
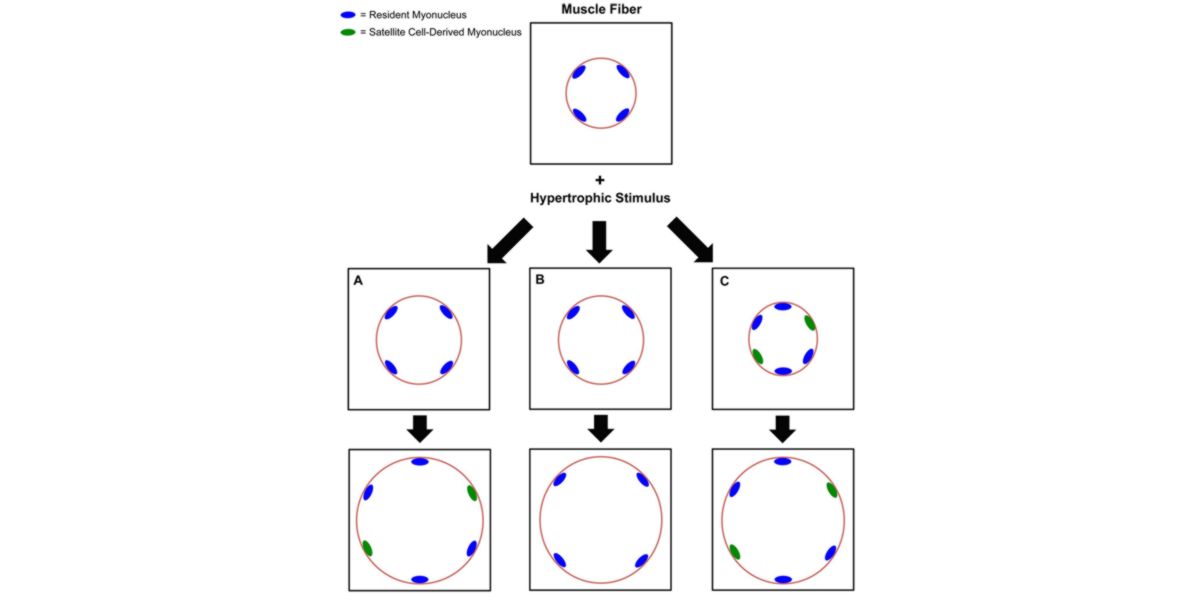
Figure IV. 3 possible phenotypes of muscular hypertrophy after a stimulus (training).
As if it were a construction in a circular perimeter, and each blue dot a worker in charge of building a certain area, when they start working (training), the workers get tired (muscle damage), and as they realise the amount of work, they call more co-workers.
That is, new workers who did not work on this building, but who come to help (green dots that are cores given by the satellite cells), and as there are more workers, they can work faster, coping with more work (because each one has a smaller area) and can make a bigger and more beautiful building.
That said, this is not always the way, and it can happen that 4 workers (assumed from the centre of the image) will use themselves to do the construction.
Perfectly, this is one of the principles of hypertrophy, which explains that each nucleus controls the protein synthesis activity (transcriptomics) of a zone of the cell, and the more myonuclei, the more capacity to cope with a greater impact of training stimulus, and the stronger the later protein synthesis (net) too. In other words, the more hypertrophy.
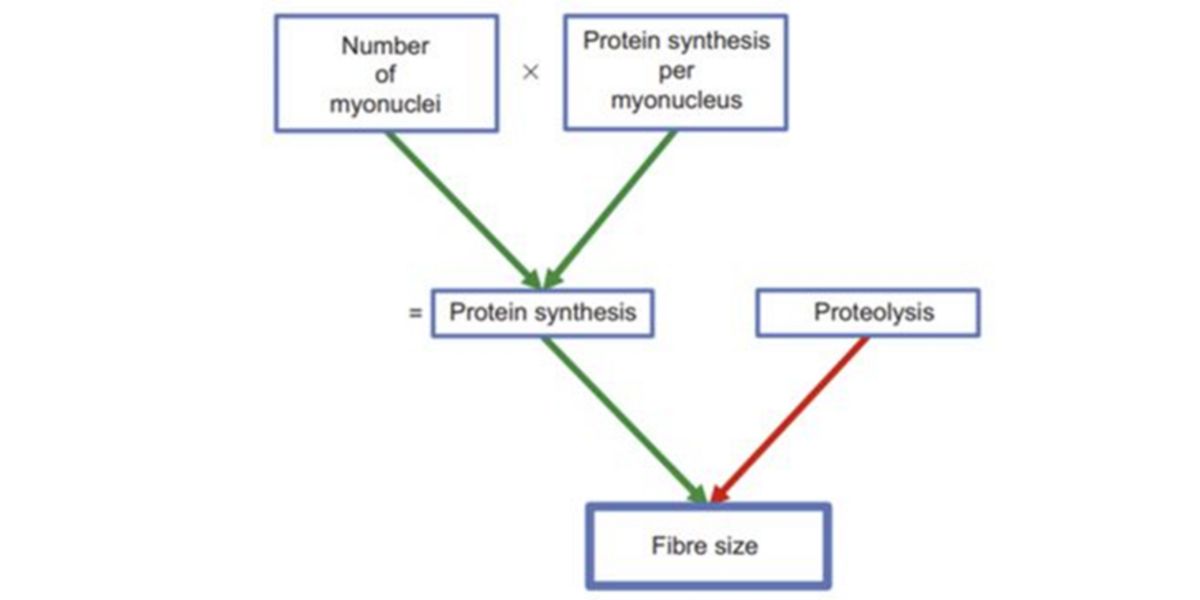
Figure V. Factors regulating fibre size (hypertrophy/atrophy)
This has been proven in several trials, where the size of the muscle fibres has been shown to have a quasi-perfect correlation with the number of myonuclei.
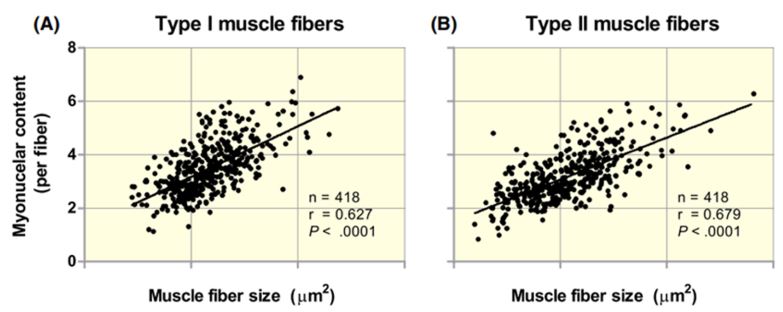
Figure VI. Relationship between muscle fibre size and number of myonuclei per fibre
Example of muscle memory
Staron et al., (1991) carried out a study on healthy women which showed that muscle memory really does exist.
The following image shows it perfectly:
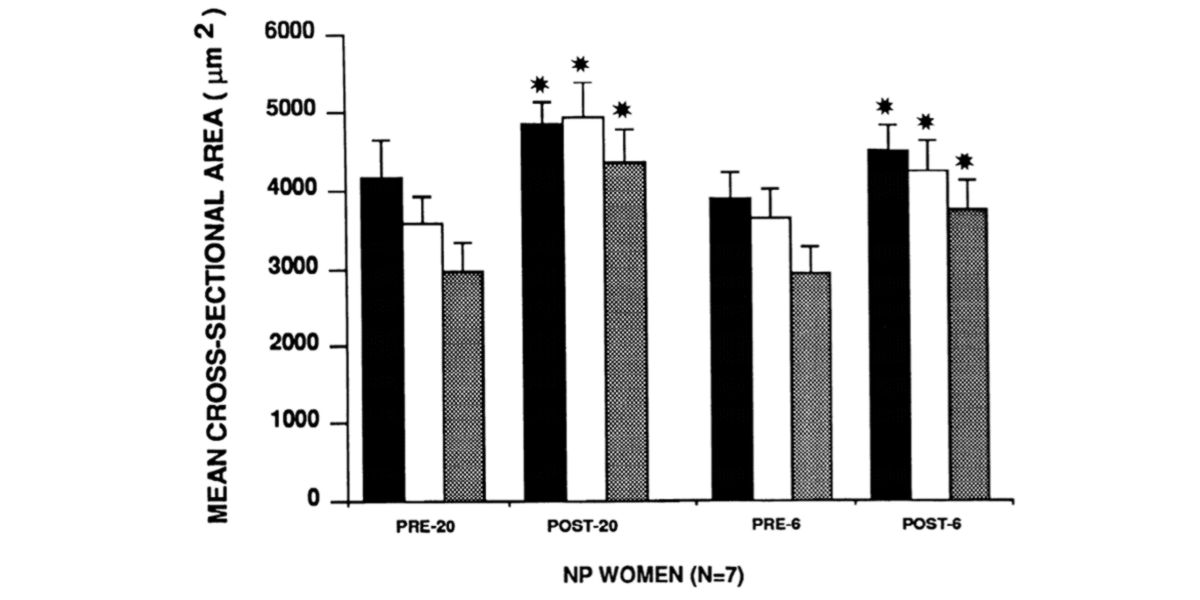
Figure VII. Changes in fibre size type I (black bars) and type II (which doesn’t matter because the classification currently used is obsolete), before and after training and retraining.
The women in their initial state (Pre-20) underwent 20 weeks of strength training (Post-20) and, as can be seen, increased the size of all their muscle fibre types; after this, they underwent 30-32 weeks of detraining, where, as can be seen, their muscle fibres, especially those that are more glycolytic (type II), decreased in size (pre-6), after which they underwent a 6-week retraining programme (post-6), with which they recovered their previous state.
Theory of myonuclear permanence
It used to be thought that during the training period, the myonuclei fused into the muscle cells by training went through an apoptotic process, i.e. as they were no longer needed, they were lost.
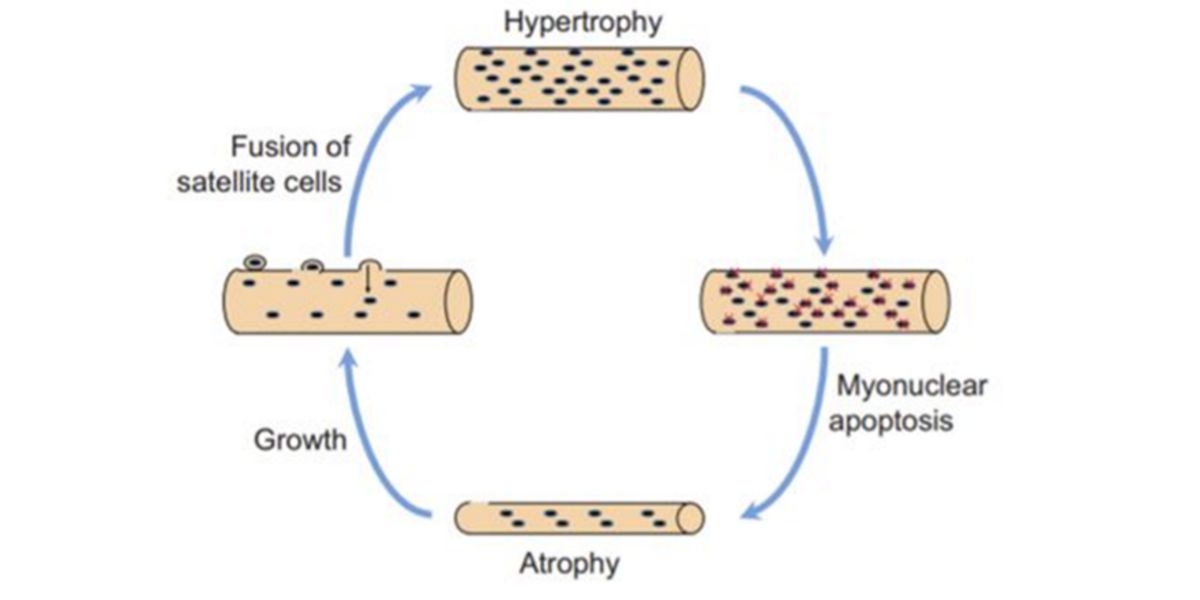
Figure VIII. Original detraining-retraining process hypothesis.
Subsequently, it was observed in in-vivo animal tests that de-training did not reduce the number of myonuclei:
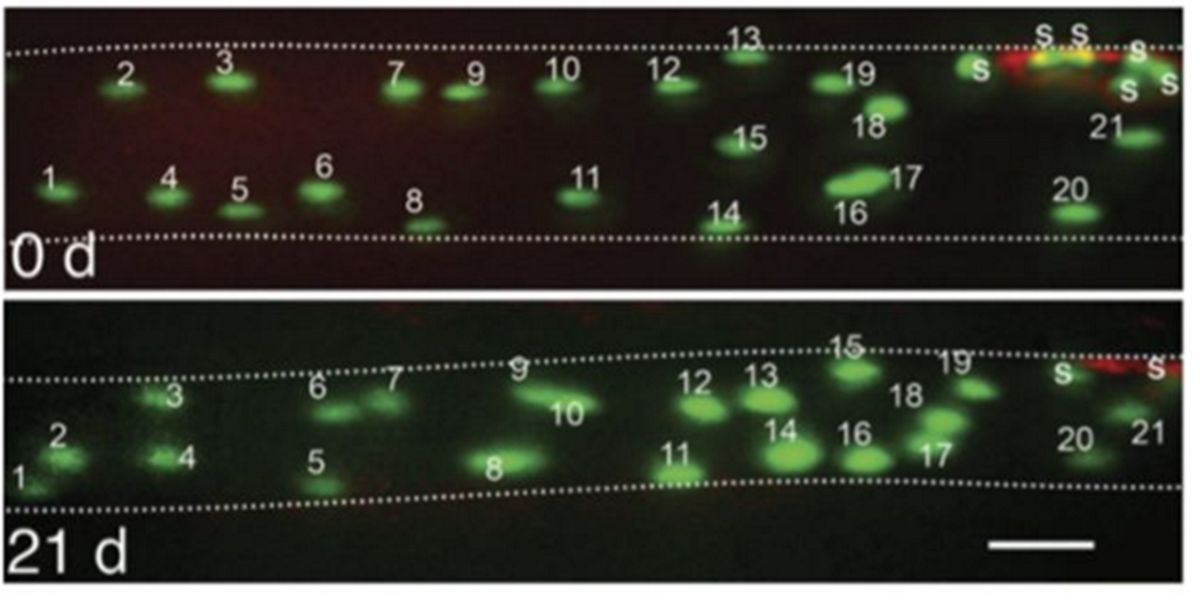
Figure IX. In vivo image of the number of myonuclei before and after 21 days of distraining in a muscle cell of an animal model.
This gave a logical justification to the muscle memory theory: we gain muscle mass faster because we do not have to invest time in the process of satellite cell migration to increase the number of myonuclei, it’s already done.
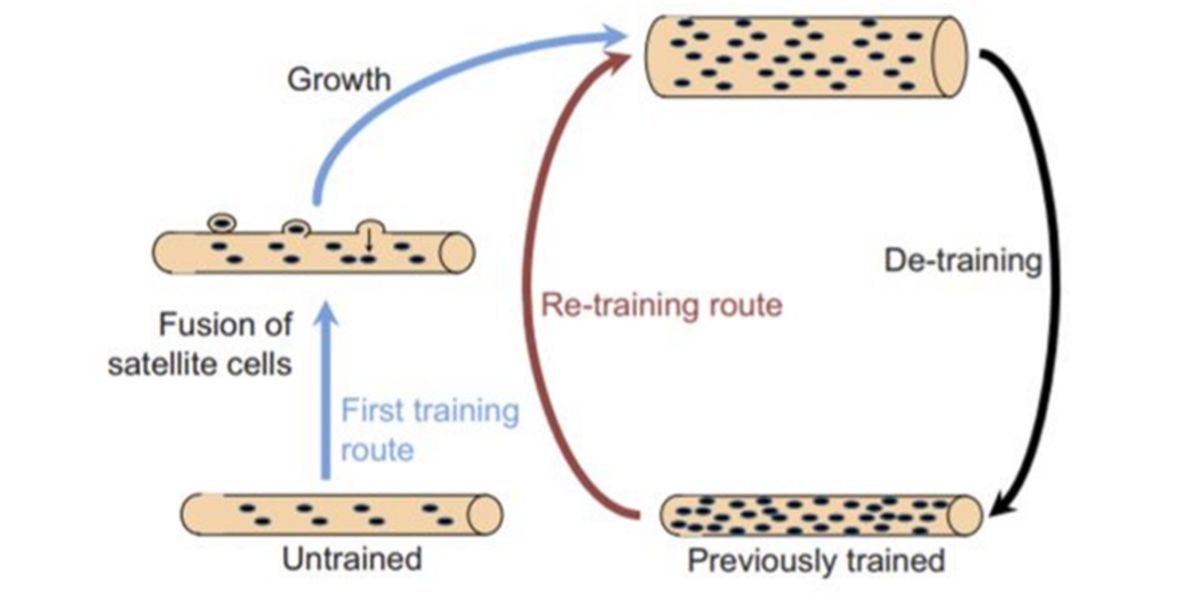
Figure X. Dominance/permanence of myonuclei hypothesis.
And that’s where the theory came from, which explains that muscle fibres decrease in size, but not in the number of nuclei gained. So, when the demands of force production increase again, the tissue recovers its previous training volume without the great efforts that are required initially.
This theory would be perfect – in fact, it was the one I had proposed as an explanation in the first article – if it were not for the fact that it was not validated in humans, and the only in-vivo study that currently exists with humans (Psilander et al., 2019) has failed to find changes in myonucleus content during the first period of training, de-training or retraining.
There goes that idea.
In addition, animal models are very heterogeneous, due to the variety of analytical methods used to discriminate true myonuclei from other cellular structures (such as fibroblasts or satellite cells), which can alter the results.
Full details of this critical analysis can be found in the review by Snijders et al. (2019).
What I mean is that according to the biological teleology, the mixed results in rodents, and the absence of trials that demonstrate this theory in humans (while there are trials that show that the content of myonuclei is not stable throughout life):
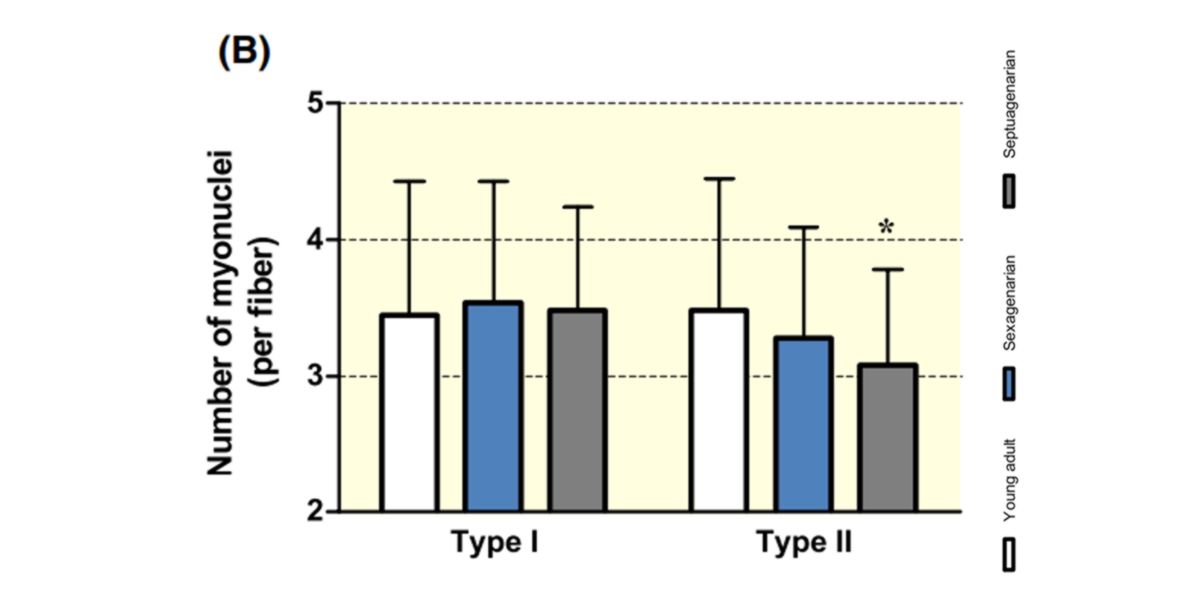
Figure XI. Changes in the number of myonuclei per fibre in young adults, sexagenarians and septagenarian subjects. Age decreases the number of myonuclei.
Epigenetic theory
In the first article I wrote, I mentioned it, but I didn’t go too far into this possibility because of the complexity of the subject. Even so, it’s currently (perhaps) the theory that presents more evidence explaining, at least, part of the reasons that underlie muscular memory in humans.
You’ll likely have heard that smoking increases the risk of lung cancer: this is an epigenetic induction process, because by exposure to pollutants present in cigarette smoke, the body undergoes a series of genomic changes that cause you to develop a pathology.
And what if strength training produced genomic changes? Of course it does!
Load training is capable of modulating the transcriptomical response of muscle tissue cells, amongst others, to increase protein synthesis so that they can become larger and exert more force (as explained above).
Some genes require exposure to the stimulus (training) for longer to undergo changes in their activation (methylation), while others may not be altered, or we have not yet been able to demonstrate this.
These changes in certain genes modulate the response of a large number of signalling pathways.
In fact, Seaborne et al. (2018)’s study shows that strength-training-induced hypomethylation of certain tested gene sets is associated with increased transcriptional activity of the PI3K-AKT-mTORC1 pathway.
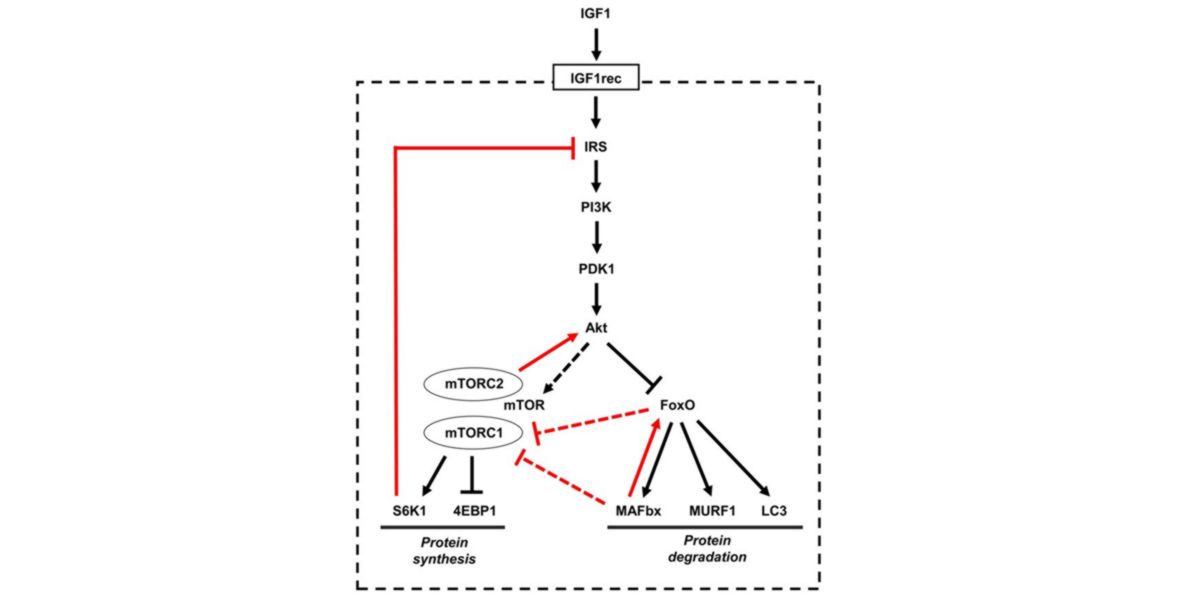
Figure XII. Metabolic pathway regulating protein turnover by activation of IGF-1 receiver and IRS-1/PI3K/Akt cascade
This means:
The article by Seaorne et al. (2018) showed that when they subjected a group of men to a 7-week training program, they increased their muscle mass.
By the time they stopped training for 7 weeks, they lost practically all of it, and when they returned to training for another 7 weeks, they recovered and surpassed their previous best form.
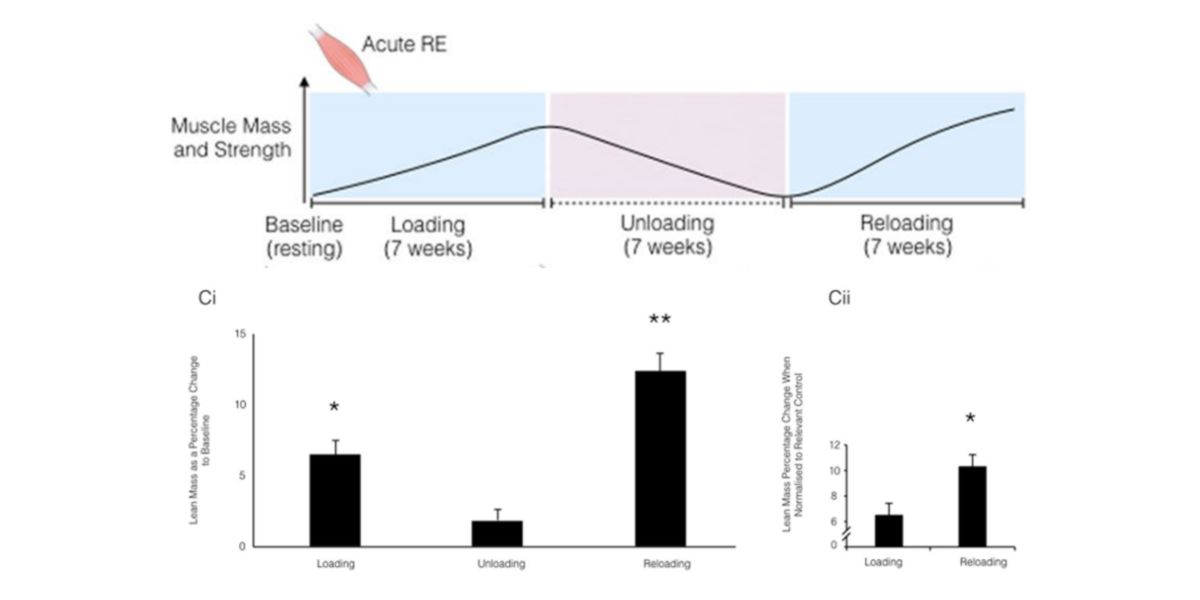
Figure XIII. Graphic representation of protocola nd results of the study.
The authors evaluated the genetic changes that occurred during the study and observed a very interesting trend that a large number of the CpGs evaluated (regions of certain genes) were hypomethylated, something that is presumed to increase the activity of the gene.
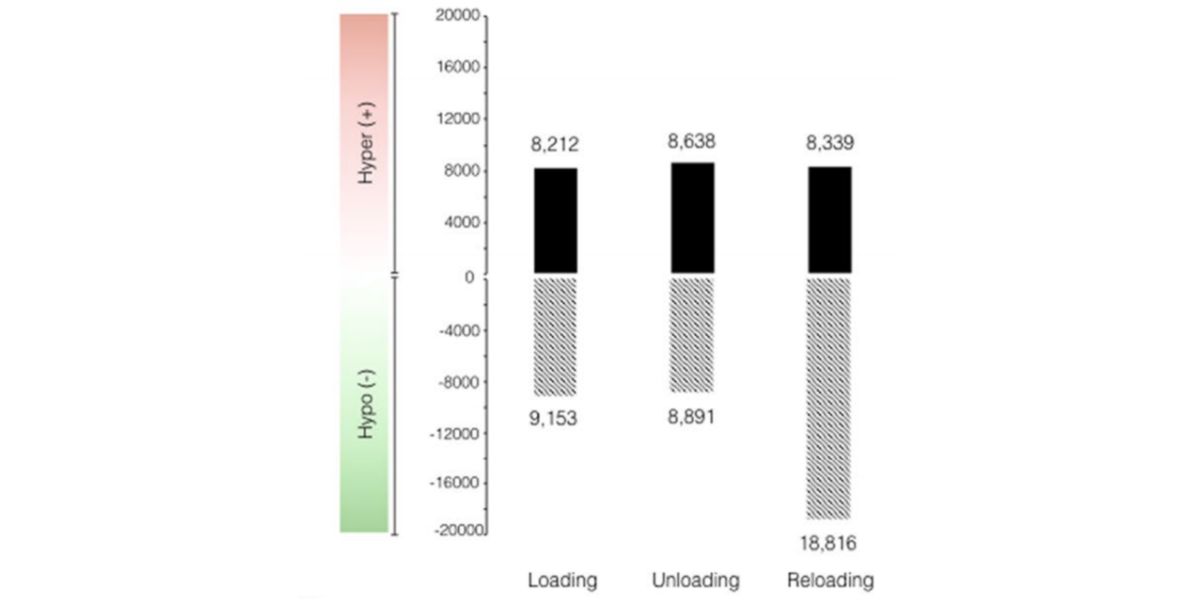
Figure XIII. Changes in the methylation of CpGs after the first period of training, detraining and retraining
As you can see in the image, after the first training period 17365 CpGs (of the 850,000 evaluated) they suffered changes in the methylation, most of them hypomethylation.
This remained practically intact during the period of detraining, and increased drastically in the period of retraining, until reaching the 18816 hypomethylated CpGs.
The authors observed that there were groups of genes (and specific genes) that showed a tendency to vary their degree of methylation depending on whether or not the subjects trained (cluster A):
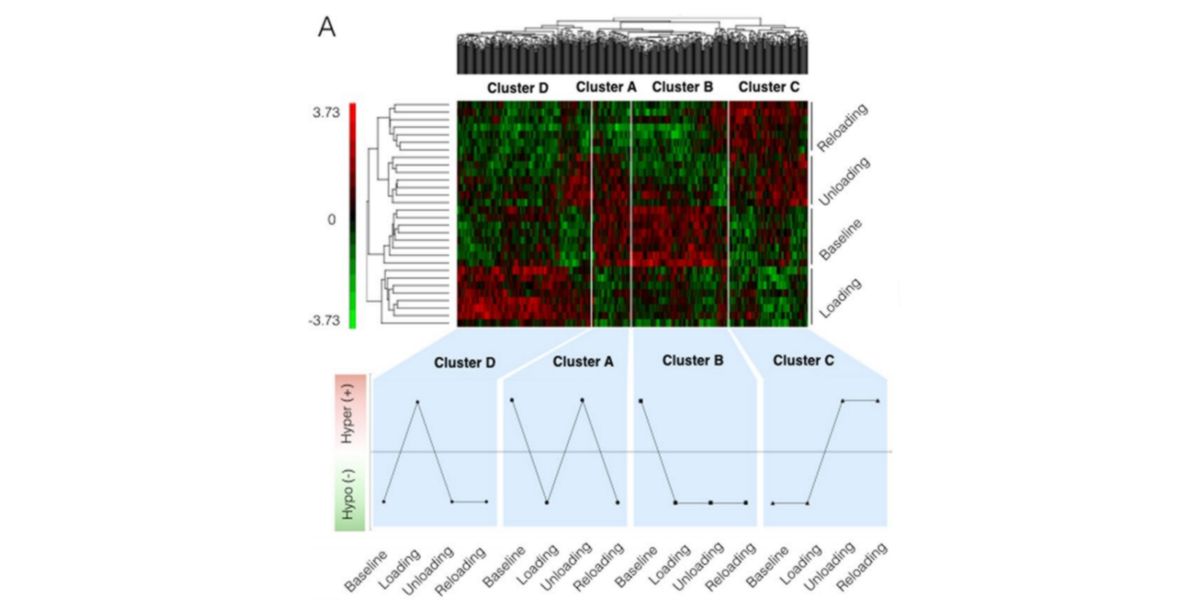
Figure XIV. Heat map and graph showing changes in the methylation of clusters of CpGs (genetic segments) at different stages of the study. Red is hyper-methylated, green hypomethylated, black no information.
While some varied acutely with training and then normalised (Cluster D), others had a late response (Cluster C) and others still changed with exposure to training and were maintained throughout the process (Cluster B).
As the behaviour of all the loci is not identical (you can see in the colour map of the image), the authors established that certain tendencies to hypo- or hyper-methylation genomic clusters were associated with the hypertrophic response of the skeletal muscle, and that this was the reason why the subjects of the study recovered their muscle mass so quickly.
That is, the authors claimed that the changes that training induced in the activity of genes (and that some of them like RPL35a / UBR5/ SETDF3 and PLA2G16 were especially sensitive to this) were unequivocally related to the increase in muscle volume and explained the reasons for the greater effects through retraining.
In sum
Although muscle memory undoubtedly exists in animal models, and appears (at least in several studies) to exist in humans, the reasons why this occurs are far from clear.
Although in rodents the theory of the myonuclear domain (permanence of myonuclei) may be a real possibility in certain circumstances, It remains to be clarified exactly which ones through standardisation of the techniques used to avoid errors in the results, and then to see if that is replicable in humans.
We also need to evaluate the epigenetic theory (which is currently the most valid in humans) on much larger samples, as the study by Seaorne et al. (2018) uses only 8 adults, and all methylations and increases in transcriptional activity of the entire genome should be sought, in addition to investigating the effects they have on the body and the magnitude of these, to determine if the relationship is really causal.
Bibliographic References
- Bruusgaard, J. C., Johansen, I. B., Egner, I. M., Rana, Z. A., & Gundersen, K. (2010). Myonuclei acquired by overload exercise precede hypertrophy and are not lost on detraining. Proceedings of the National Academy of Sciences of the United States of America, 107(34), 15111–15116.
- Gundersen, K. (2016). Muscle memory and a new cellular model for muscle atrophy and hypertrophy. Journal of Experimental Biology, 219(2), 235–242.
- Hawke, T. J., & Garry, D. J. (2001). Myogenic satellite cells: Physiology to molecular biology. Journal of Applied Physiology, 91(2), 534–551.
- Murach, K. A., Englund, D. A., Dupont-Versteegden, E. E., McCarthy, J. J., & Peterson, C. A. (2018). Myonuclear domain flexibility challenges rigid assumptions on satellite cell contribution to skeletal muscle fiber hypertrophy. Frontiers in Physiology, 9(MAY), 635.
- Ryan, B. J., Wachsmuth, N. B., Schmidt, W. F., Byrnes, W. C., Julian, C. G., Lovering, A. T., … Roach, R. C. (2014). Altitudeomics: Rapid hemoglobin mass alterations with early acclimatization to and de-acclimatization from 5260 m in healthy humans. PLoS ONE, 9(10), e108788.
- Schiaffino, S., & Mammucari, C. (2011). Regulation of skeletal muscle growth by the IGF1-Akt/PKB pathway: Insights from genetic models. Skeletal Muscle, 1(1), 4.
- Seaborne, R. A., Strauss, J., Cocks, M., Shepherd, S., O’Brien, T. D., Van Someren, K. A., … Sharples, A. P. (2018). Human Skeletal Muscle Possesses an Epigenetic Memory of Hypertrophy. Scientific Reports, 8(1), 1898.
- Snijders, T., Aussieker, T., Holwerda, A., Parise, G., van Loon, L. J. C., & Verdijk, L. B. (2020). The concept of skeletal muscle memory: Evidence from animal and human studies. Acta Physiologica, e13465.
- Staron, R. S., Leonardi, M. J., Karapondo, L., Malicky, E. S., Falkel, J. E., Hagerman, F. C., & Hikida, R. S. (1991). Strength and skeletal muscle adaptations in heavy-resistance-trained women after detraining and retraining. Journal of Applied Physiology, 70(2), 631–640.
Related Entries
- What is Sarcopenia?
- Protein Synthesis: Everything You Need to Know and How to Improve It
- Anabolic Pathways: Protein Biosynthesis



 Fitness, Nutrition, Health and Sports Blog In the HSNstore Blog you will find tips about Fitness, sport in general, nutrition, and health – HSNstore.com
Fitness, Nutrition, Health and Sports Blog In the HSNstore Blog you will find tips about Fitness, sport in general, nutrition, and health – HSNstore.com 




